
Batteries achieve the desired operating voltage by connecting several cells in series; each cell adds its voltage potential to arrive at the total terminal voltage. Parallel connection attains higher capacity by adding up the total ampere-hour (Ah).
Some packs may consist of a combination of series and parallel connections. Laptop batteries commonly have four 3,6 V Li-ion cells in series to achieve a nominal voltage 14,4 V, and two in parallel to boost the capacity from 2400 mAh to 4800 mAh. Such a configuration is called 4S2P, meaning four cells in series and two in parallel. Insulating foil between the cells prevents the conductive metallic skin from causing an electrical short.
Most battery chemistries lend themselves to series and parallel connection. It is important to use the same battery type with equal voltage and capacity (Ah) and never to mix different makes and sizes. A weaker cell would cause an imbalance. This is especially critical in a series configuration because a battery is only as strong as the weakest link in the chain. An analogy is a chain in which the links represent the cells of a battery connected in series (Figure 1).

A weak cell may not fail immediately but will get exhausted more quickly than the strong ones when on a load. On charge, the low cell fills up before the strong ones because there is less to fill and it remains in over-charge longer than the others. On discharge, the weak cell empties first and gets hammered by the stronger brothers. Cells in multi-packs must be matched, especially when used under heavy loads.
Single-cell applications
The single-cell configuration is the simplest battery pack; the cell does not need matching and the protection circuit on a small Li-ion cell can be kept simple. Typical examples are mobile phones and tablets with one 3,60 V Li-ion cell. Other uses of a single cell are wall clocks, which typically use a 1,5 V alkaline cell, wristwatches and memory backup, most of which are very low-power applications.
Series connection
Portable equipment needing higher voltages use battery packs with two or more cells connected in series. Figure 2 shows a battery pack with four 3,6 V Li-ion cells in series, also known as 4S, to produce 14,4 V nominal. In comparison, a six-cell lead acid string with 2 V/cell will generate 12 V, and four alkaline with 1,5 V/cell will give 6 V.
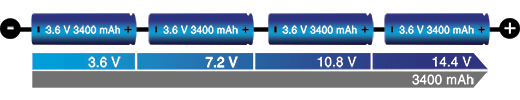
If you need an odd voltage of, say, 9,50 volts, connect five lead acid, eight NiMH or NiCd, or three Li-ion in series. The end battery voltage does not need to be exact as long as it is higher than what the device specifies. A 12 V supply might work in lieu of 9,50 V. Most battery-operated devices can tolerate some over-voltage; the end-of-discharge voltage must be respected, however.
Parallel connection
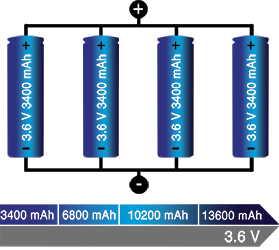
If higher currents are needed and larger cells are not available or do not fit the design constraint, one or more cells can be connected in parallel. Most battery chemistries allow parallel configurations with little side effect. Figure 3 illustrates four cells connected in parallel in a P4 arrangement. The nominal voltage of the illustrated pack remains at 3,60 V, but the capacity (Ah) and runtime are increased fourfold.
A cell that develops high resistance or opens is less critical in a parallel circuit than in a series configuration, but a failing cell will reduce the total load capability. It’s like an engine only firing on three cylinders instead of on all four. An electrical short, on the other hand, is more serious as the faulty cell drains energy from the other cells, causing a fire hazard. Most so-called electrical shorts are mild and manifest themselves as elevated self-discharge.
A total short can occur through reverse polarisation or dendrite growth. Large packs often include a fuse that disconnects the failing cell from the parallel circuit if it were to short. Figure 4 illustrates a parallel configuration with one faulty cell.
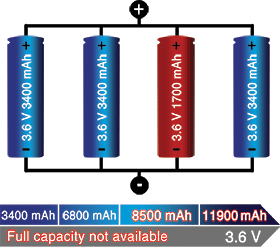
A weak cell will not affect the voltage but provide a low runtime due to reduced capacity. A shorted cell could cause excessive heat and become a fire hazard. On larger packs a fuse prevents high current by isolating the cell.
Series/parallel connection
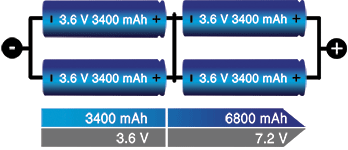
The series/parallel configuration shown in Figure 5 enables design flexibility and achieves the desired voltage and current ratings with a standard cell size. The total power is the product of voltage times current; four 3,6 V (nominal) cells multiplied by 3400 mAh produce 12,24 Wh. Four 18650 energy cells of 3400 mAh each can be connected in series and parallel as shown to get 7,2 V nominal and 12,24 Wh. The slim cell allows flexible pack design but a protection circuit is needed.
Li-ion lends itself well to series/parallel configurations but the cells need monitoring to stay within voltage and current limits. Integrated circuits (ICs) for various cell combinations are available to supervise up to 13 Li-ion cells. Larger packs need custom circuits, and this applies to e-bike batteries, hybrid cars and the Tesla Model 85 that devours over 7000 18650 cells to make up the 90 kWh pack.
Terminology to describe series and parallel connection
The battery industry specifies the number of cells in series first, followed by the cells placed in parallel. An example is 2S2P. With Li-ion, the parallel strings are always made first; the completed parallel units are then placed in series. Li-ion is a voltage-based system that lends itself well for parallel formation. Combining several cells into a parallel and then adding the units serially reduces complexity in terms of voltage control for pack protection.
Building series strings first and then placing them in in parallel may be more common with NiCd packs to satisfy the chemical shuttle mechanism that balances charge at the top of charge. 2S2P is common; white papers have been issued that refer to 2P2S when a serial string is paralleled.
Safety devices in series and parallel connection
Positive temperature coefficient (PTC) switches and charge interrupt devices (CID) protect the battery from over-current and excessive pressure. While recommended for safety in a smaller 2- or 3-cell pack with serial and parallel configuration, these protection devices are often being omitted in larger multi-cell batteries, such as those for power tools.
The PTC and CID work as expected to switch off the cell on excessive current and internal cell pressure; however the shutdown occurs in cascade format. While some cells may go offline early, the load current causes excess current on the remaining cells. Such an overload condition could lead to a thermal runaway before the remaining safety devices activate.
For more information contact Michael Rogers, Uniross Batteries, +27 (0)11 466 1156, [email protected], www.uniross.co.za
| Tel: | +27 11 466 1156 |
| Email: | [email protected] |
| www: | www.uniross.co.za |
| Articles: | More information and articles about Uniross Batteries |

© Technews Publishing (Pty) Ltd | All Rights Reserved