
As batteries were beginning to be mass-produced, the jar design changed to the cylindrical format. The large F cell for lanterns was introduced in 1896 and the D cell followed in 1898. With the need for smaller cells, the C cell followed in 1900, and the popular AA was introduced in 1907.
Other formats have been introduced in the years since, and this article compares the pros and cons of the commonly used cylindrical, button, prismatic and pouch cell formats.
Cylindrical cell
The cylindrical cell continues to be one of the most widely used packaging styles for primary and secondary batteries. The advantages are ease of manufacture and good mechanical stability. The tubular cylinder can withstand high internal pressures without deforming.
Many lithium and nickel-based cylindrical cells include a positive thermal coefficient (PTC) switch. When exposed to excessive current, the normally conductive polymer heats up and becomes resistive, stopping current flow and acting as short circuit protection. Once the short is removed, the PTC cools down and returns to the conductive state.
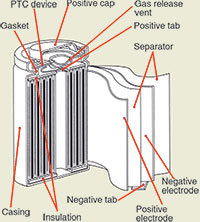
Most cylindrical cells also feature a pressure relief mechanism, and the simplest design utilises a membrane seal that ruptures under high pressure. Leakage and dry-out may occur after the membrane breaks. Re-sealable vents with a spring-loaded valve are the preferred design. Some consumer Li-ion cells include the charge interrupt device (CID) that physically and irreversibly disconnects the cell when activated by an unsafe pressure build-up. Figure 1 shows a cross section of a cylindrical cell.
The cylindrical cell design has good cycling ability, offers a long calendar life, and is economical, but it is heavy and has low packaging density due to space cavities. Typical applications for the cylindrical cell are power tools, medical instruments, laptops and e-bikes.
To allow variations within a given size, manufacturers use partial cell lengths, such as half and three-quarter formats, and nickel-cadmium provides the largest variety of cell choices. Some spilled over to nickel-metal-hydride, but not to lithium-ion as this chemistry established its own formats.
The 18650 remains one of the most popular cell packages. Typical applications for the 18650 Li-ion are power tools, medical devices, laptops and e-bikes. The metallic cylinder measure18 mm in diameter and 65 mm in length, while the larger 26650 cell measures 26 mm in diameter.
In 2013, 2,55 billion 18650 cells were produced. Early energy cells had 2,2 Ah capacity, which was then superseded by the 2,8 Ah cell. New cells have reached 3,4 Ah, and cell manufacturers are preparing for 3,9 Ah models in the future.
The 18650 could well be the most optimised cell; it offers one of the lowest costs per Wh and has good reliability records. As consumers move to the flat designs in smartphones and tablets, the demand for the 18650 is fading and Figure 2 shows the over-supply that is being corrected thanks to the demand of the Tesla electric vehicles that also use this cell format for now. As of the end of 2016, the battery industry fears battery shortages to meet the growing demand for electric vehicles.
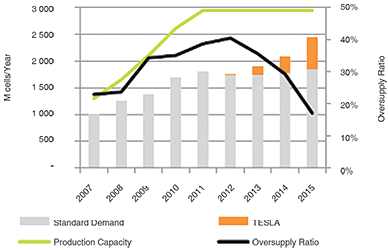
The demand for the 18650 would have peaked in 2011 had it not been for new demands in military, medical and drones, as well as the Tesla electric car. The switch to a flat design in consumer products and larger format for the electric powertrain will eventually saturate the 18650.
There are other cylindrical Li-ion formats with dimensions of 20700, 21700 and 22700. Meanwhile, Tesla, Panasonic and Samsung have decided on the 21700 for ease of manufacturing, optimal capacity and other benefits. While the 18650 has a volume of approximately 16 cm³ with a capacity of around 3000 mAh, the 21700 cell has approximately 24 cm³ with a capacity of up to 6000 mAh, essentially doubling the capacity with a 50% increase in volume.
Tesla Motor refers to the company’s new 21700 as the ‘highest energy density cell that is also the cheapest.’ (The 2170 nomenclature Tesla advocates is not totally correct; the last zero of the 21700 model describes a cylindrical cell harmonising with the IEC standard.)
The larger 26650 cell with a diameter of 26 mm does not enjoy the same popularity as the 18650. The 26650 is commonly used in load-levelling systems. A thicker cell is said to be harder to build than a thinner one, so making the cell longer is preferred. There is also a 26700 made by E-One Moli Energy.
Some lead-acid systems also borrow the cylindrical design. Known as the Hawker Cyclone, this cell offers improved cell stability, higher discharge currents and better temperature stability compared to the conventional prismatic design. The Hawker Cyclone has its own format.
Even though the cylindrical cell does not fully utilise the space by creating air cavities on side-by-side placement, the 18650 has a higher energy density than a prismatic/pouch Li-ion cell. The 3 Ah 18650 delivers 248 Ah/kg, whereas a modern pouch cell has about 140 Ah/kg. The higher energy density of the cylindrical cell compensates for its less ideal stacking abilities, and the empty space can always be used for cooling to improve thermal management.
Cell disintegration cannot always be prevented but propagation can. Cylindrical cells are often spaced apart to stop propagation should one cell take off. In addition, a cylindrical design does not change size. By comparison, a 5 mm prismatic cell can expand to 8 mm with use, and allowances must be made.
Button cell
The button cell, also known as coin cell, enabled compact design in portable devices of the 1980s. Higher voltages were achieved by stacking the cells into a tube. Cordless telephones, medical devices and security wands at airports used these batteries.
Although small and inexpensive to build, the stacked button cell fell out of favour and gave way to more conventional battery formats. A drawback of the button cell is swelling if charged too rapidly. Button cells have no safety vent and can only be charged at a 10- to 16-hour charge; however, newer designs claim rapid charge capability.
Most button cells in use today are non-rechargeable and are found in medical implants, watches, hearing aids, car keys and memory backup.
Prismatic cell
Introduced in the early 1990s, the modern prismatic cell satisfies the demand for thinner sizes. Wrapped in elegant packages resembling a box of chewing gum or a small chocolate bar, prismatic cells make optimal use of space by using the layered approach. Other designs are wound and flattened into a pseudo-prismatic jelly roll.
These cells are predominantly found in mobile phones, tablets and low-profile laptops ranging from 800 mAh to 4 000 mAh. No universal format exists and each manufacturer designs its own.
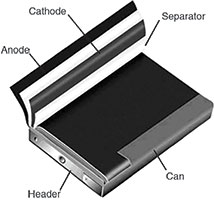
Prismatic cells are also available in large formats. Packaged in welded aluminium housings, the cells deliver capacities of 20–50 Ah and are primarily used for electric powertrains in hybrid and electric vehicles. Figure 3 shows the prismatic cell.
The prismatic cell improves space utilisation and allows flexible design, but it can be more expensive to manufacture, less efficient in thermal management and have a shorter cycle life than the cylindrical design. Allowance needs to be made for some swelling.
The prismatic cell requires a firm enclosure to achieve compression. Some swelling due to gas build-up is normal, and growth allowance must be made; a 5 mm cell can grow to 8 mm after 500 cycles. Use of the battery should be discontinued if the distortion presses against the battery compartment – bulging batteries can damage equipment and compromise safety.
Pouch cell

In 1995, the pouch cell surprised the battery world with a radical new design. Rather than using a metallic cylinder and glass-to-metal electrical feed-through, conductive foil-tabs were welded to the electrodes and brought to the outside in a fully sealed way. Figure 4 illustrates a pouch cell.
The pouch cell offers a simple, flexible and lightweight solution to battery design. Some stack pressure is recommended but allowance for swelling must be made. The pouch cell can deliver high load currents but it performs best under light loading conditions and with moderate charging.
The pouch cell makes most efficient use of space and achieves 90–95% packaging efficiency, the highest among battery packs. Eliminating the metal enclosure reduces weight, but the cell needs support and allowance to expand in the battery compartment. The pouch packs are used in consumer, military and automotive applications. No standardised pouch cells exist; each manufacturer designs its own.
Pouch packs are commonly Li-polymer. Small cells are popular for portable applications requiring high load currents, such as drones and hobby gadgets. The larger cells in the 40 Ah range serve in energy storage systems because fewer cells simplify the battery design.
Although easily stackable, provision must be made for swelling. While smaller pouch packs can grow 8–10% over 500 cycles, large cells may expand to that size in 5000 cycles. It is best not to stack pouch cells on top of each other but to lay them flat, side by side or allow extra space in between them. Avoid sharp edges that can stress the pouch cells as they expand.
Extreme swelling is a concern. Users of pouch packs have reported up to 3% swelling incidents on a poor batch run. The pressure created can crack the battery cover, and in some cases, break the display and electronic circuit boards. Discontinue using an inflated battery and do not puncture the bloating cell in close proximity to heat or fire, as the escaping gases can ignite.
Swelling can occur due to gassing, but improvements are being made with newer designs. Large pouch cell designs experience less swelling. The gases contain mainly CO2 (carbon dioxide) and CO (carbon monoxide).
Pouch cells are manufactured by adding a temporary ‘gasbag’ on the side. Gases escape into the gasbag while forming the solid electrolyte interface (SEI) during the first charge. The gasbag is cut off and the pack is resealed as part of the finishing process. Forming a solid SEI is key to good formatting practices.
Subsequent charges should produce minimal gases, however, gas generation, also known as gassing, cannot be fully avoided. It is caused by electrolyte decomposition as part of usage and ageing, and stresses such as overcharging and overheating promote gassing. Ballooning with normal use often hints at a flawed batch.
The technology has matured and prismatic and pouch cells have the potential for greater capacity than the cylindrical format. Large flat packs serve electric powertrains and energy storage systems with good results.
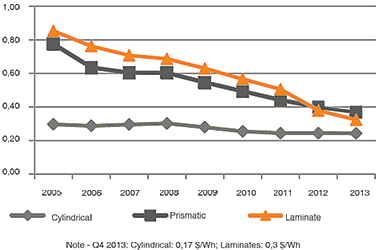
The cost per kWh in the prismatic/pouch cell is still higher than with the 18650 cell, but this is changing. Figure 5, compares the price of the cylindrical, prismatic and pouch cells, also known as laminated. Flat-cell designs are getting price competitive and battery experts predict a shift towards these cell formats, especially if the same performance criteria of the cylindrical cell can be met.
For more information contact Michael Rogers, Uniross Batteries, +27 11 466 1156, [email protected], www.uniross.co.za
| Tel: | +27 11 466 1156 |
| Email: | [email protected] |
| www: | www.uniross.co.za |
| Articles: | More information and articles about Uniross Batteries |

© Technews Publishing (Pty) Ltd | All Rights Reserved