
What causes a battery to wear down - is it mechanical or chemical? The answer is 'both'. A battery is a perishable product that starts deteriorating right from the time it leaves the factory. Similar to a spring under tension, a battery seeks to revert back to its lowest denominator.
The speed of ageing is subject to depth of discharge, environmental conditions, charge methods and maintenance procedures, or the lack thereof. Ageing and user-conditions affect each battery chemistry differently.
As part of an ongoing research programme to find the optimum battery system for wireless applications, Cadex has performed life cycle tests on nickel cadmium (NiCd), nickel metal hydride (NiMH) and lithium ion (Li-ion) batteries. All tests were carried out on the Cadex C7000 battery analysers in the test labs of Cadex Electronics in Vancouver, Canada.
The batteries received an initial full-charge, and then underwent a regime of continued discharge/charge cycles. The internal resistance was measured with Cadex's proprietary 'OhmTest' method, and the self-discharge was obtained from time-to-time by reading the capacity loss incurred during a 48-hour rest period. The test programme involves 53 batteries of different models and chemistries.
When conducting battery tests in a laboratory, it should be noted that the performance in a protected environment is commonly superior to those in field use. Elements of stress and inconsistency that are present in everyday use cannot always be simulated accurately in the lab.
Battery performance as a function of cycling
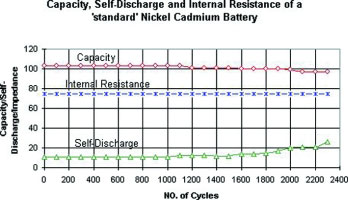
In terms of life cycling, the standard NiCd is the most enduring battery. In Figure 1 we examine the capacity, internal resistance and self-discharge of a 7,2 V, 900 mA NiCd battery with standard cells. Due to time constraints, the test was terminated after 2200 cycles. During this period, the capacity remains steady, the internal resistance stays flat at 75 mΩ and the self-discharge is stable. This battery receives a grade 'A' for almost perfect performance.
The readings on an ultra-high capacity NiCd are less favourable but still better than other chemistries in terms of endurance. Although up to 60% higher in energy density than the standard NiCd version, Figure 2 shows a steady drop of capacity during the 2000 cycles delivered. At the same time, the internal resistance rises slightly. A more serious degradation is the increase of self-discharge after 1000 cycles. This deficiency manifests itself in shorter runtimes because the battery consumes some energy, even if not in use.
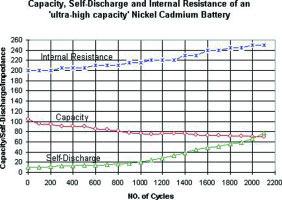
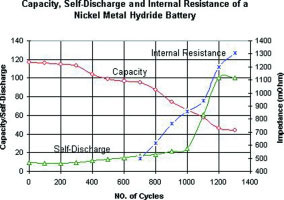
Figure 3 examines the NiMH, a battery that offers high energy density at low cost. We observe good performance at first but past the 300-cycle mark, the performance starts to drift downwards rapidly. One can observe the swift increase in internal resistance and self-discharge after cycle count 700.
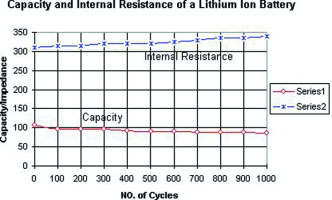
The Li-ion battery offers advantages that neither the NiCd nor NiMH can meet. In Figure 4 we examine the capacity and internal resistance of a typical Li-ion. A gentle and predictable capacity drop is observed over 1000 cycles and the internal resistance increases only slightly. Because of low readings, self-discharge has been omitted on this test.
Is the Li-ion truly superior?
Today's battery research focuses heavily on lithium systems, so much so that one could assume that all future applications will be lithium-based. This lithium hype is especially apparent when attending battery conferences where battery manufacturers from all corners of the world meet to present their latest achievements. Just a few years ago, the argument was NiCd against NiMH; then the speakers argued on the benefit of Li-ion over NiMH and today, the emphasis is on Li-ion polymer. The nickel cadmium (NiCd) and nickel metal hydride (NiMH) are hardly mentioned.
In many aspects, the Li-ion is superior to nickel or lead-based batteries. There is one weak point with the Li-ion that, for unknown reasons, is seldom mentioned by the battery manufacturer. It is ageing. Capacity deterioration is noticeable after one year, whether the battery is in storage or use. Past two years, the battery frequently fails.
Although less of a concern in the fast moving cellphone and notebook market, storage at operational readiness is an important factor in most industrial applications. In Table 1 we examine the capacity loss as a function of charge level and storage temperature.

It is not recommended to keep Li-ion batteries in storage for a long time. Instead, the packs should be rotated like perishable food. The buyer should be aware of the manufacturing date when purchasing a replacement Li-ion battery. Unfortunately, this information is often encoded in an encrypted serial number and is only available to the manufacturer. The recommended charge level is 40% and the storage temperature should be 15°C or less.
Summary
Research has brought about a variety of battery chemistries, each offering distinct advantages but none providing a fully satisfactory solution. However, with today's selection of battery systems, better choices can be made to tailor to specific user applications.
Relentless downsizing has pressured manufacturers to invent smaller battery sizes. By packing more energy into a pack, other qualities may be neglected, one of which is longevity. Long service life and predictable low mΩ reading are found in the NiCd family. However, this chemistry is being replaced, where applicable, with systems containing higher energy density. In addition, negative publicity about the memory phenomenon and concerns of toxicity on careless disposal, are causing equipment manufacturers to seek alternative choices.
Once hailed as the superior battery system, the NiMH has also failed to provide the universal battery solution for the twenty-first century. Shorter than expected service life remains a major complaint by users.
For a fast-moving consumer market that replaces the equipment every two years, the Li-ion battery is the best choice. However, for applications that need fully charged batteries that often must be kept in warm storage conditions, the Li-ion does pose a problem in terms of longevity. So far, the emerging lithium polymer system has been unable to overcome the shortcomings of the Li-ion. Other than lighter and thinner designs, the cost-to-energy ratio of Li-Polymer is among the highest of rechargeable batteries.
With the rapid developments in technology occurring today, battery systems may soon become viable that use neither nickel, lead nor lithium. Fuel cells, which do not rely on electro-chemical processes but allow refuelling similar to a vehicle, may solve the portable energy needs for the future. Instead of a charger, the user carries a bottle of liquid energy. Although still many years from practical reality, such a battery would truly change the way we live and work.
| Tel: | +27 11 466 1156 |
| Email: | [email protected] |
| www: | www.uniross.co.za |
| Articles: | More information and articles about Uniross Batteries |

© Technews Publishing (Pty) Ltd | All Rights Reserved