
Lead-acid batteries are widely used in telecommunication and UPS applications. It is well known that proper management and regular maintenance can extend the life of a battery, but what are the possible causes of battery failure and what procedures should be undertaken if the battery fails? The following are the common battery error scenarios and handling instructions.
Battery leakage
There are four possible causes for a lead-acid battery leakage. First, the upper cover and battery container might not be well sealed, or the sealing glue could be cracked due to impact damage. Second, the safety valve may have failed to function, resulting in acid gas leakage or acid spillage. Third, the terminal glue might not be completely hardened, which leads to the acid gas or acid electrolyte leaking around the terminals. And fourth, the battery container could be cracked or broken in places.
To determine the cause and action to be taken, the battery must be visually inspected to determine where the acid is leaking from. The top cover should be opened to see if there are any traces of acid leakage around the safety valve, following which the safety valve needs to be removed in order to check the electrolyte level inside the battery.
After completing the above steps, if no defects are found then an air tightness test should be performed. Finally, the battery needs to be charged in order to check if there are any electrolyte leaks. If the battery still leaks, the defective unit must be replaced and should not be used as it is unable to support the power load. Moreover, it may cause damage to the equipment and possible personal injury.
Battery container deformation
The charge and discharge reaction of a valve regulated lead-acid (VRLA) battery can be expressed by the following chemical reaction:
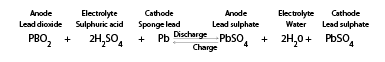
During the reaction, it causes electrolysis of the water content of the electrolyte, which generates O2 (oxygen) gas from the positive plate and H2 (hydrogen) gas from the negative plate. If oxygen and hydrogen escape, the battery will gradually dry out and eventually shorten the battery life.
The design of the VRLA battery greatly reduces the water loss during charging, where gases generated during the charge cycle are recombined in a so-called ‘oxygen cycle’. During charging, oxygen is generated from the positive plate and reacts with the sponge lead of the negative plate. When charging continues, the oxygen will recombine with the hydrogen being generated by the negative plate to form water. The water content of the electrolyte therefore remains at the same level unless the charging rate is too high.
The common charging problems can be roughly divided into two categories: undercharging and overcharging. If the charge voltage is too low or the charging time is insufficient, the current flow will stop before the battery is fully charged. Undercharging will result in gradual loss of operation time and capacity within the successive charge/discharge cycles. If the condition persists it will result in some of the lead sulphate remaining on the electrodes, resulting is the permanent loss of battery capacity.
On the other hand, overcharging will cause excessive gassing and drying out of the electrolyte, which may contribute to the risk of thermal runaway. After reaching full charge, excessive current will flow into the battery. This high charge voltage causes water decomposition in the electrolyte and the resultant premature ageing of the battery. As the battery temperature increases, so too it will accept more current and it will heat up even further. This is called thermal runaway and it will cause serious damage to the battery.
Thermal runaway may be caused by overcharging, excessively high recharge current, short circuited cells, a high ambient temperature working environment, grounding faults or a combination of these. This will ultimately lead to the battery container swelling up or deforming. Thermal runaway could also result in the inability of battery to support the power load, the emission of hydrogen sulphide, and even worse, may cause the battery to catch fire.
The user should pay careful attention to the potential risks that may be caused by thermal runaway. The faulty battery must be replaced and the cause of the thermal runaway condition corrected.
If a group of batteries is deformed at the same time, the battery voltage should be checked. If the voltage is normal, then the voltage of individual cells should be measured to determine whether they are short-circuited. If there is no short circuit, the deformation is most likely caused by thermal runaway due to overcharging.
The ambient temperature should be checked to ensure that the battery is not installed or operating in a high ambient temperature environment. Charging parameters of the battery charger must be checked to ensure that the voltage setting is not too high. If the charger is defective, it must be replaced with a charger that has a quality certificate and an overcharge protection function.
Individual battery performance lags behind
The balancing of a battery pack connected in series can be quite challenging. There will always be the possibility of under-performing batteries in the pack due to various reasons such as production flaws, raw material quality, incorrect usage or lack of maintenance.
If this occurs, the user should first charge the battery pack and then discharge it, and then measure the voltage of each battery during the discharging cycle to identify the under-performing battery which lacks discharge capacity. The under-performing battery can then be recharged, while ensuring that the battery temperature does not exceed 50°C.
If the battery capacity cannot be recovered after repeated charge/discharge procedures, the battery may have a short circuit problem, or the active material of the plates may have deteriorated. It could also be an indication that the grid has corroded and expanded, or the electrolyte has dried out, causing the battery capacity to drop and thus unable to recover. In this situation, the user should replace this battery.
On the other hand, one could find that the performance of the battery does not meet the requirements of the system, even if the battery capacity is still greater than the acceptable rated capacity. However, because the load discharge time cannot meet the minimum system requirements, the battery should not continue to be used.

© Technews Publishing (Pty) Ltd | All Rights Reserved